 
You can find this mass distribution on several internet sites. The first peak expectedly will lie between about mass numbers 90 and 100, and the second peak will fall between about mass numbers 130 and 140. The preferred mass distribution of the products is actually bimodal-i.e., if you plot the yields of fission products of given masses versus mass number, you will generate a curve with two peaks. The two major fragments, which we refer to as fission products, may have a wide spectrum of nuclear masses, characterized by the fact that the fission process does not favor an equal mass distribution between the two fragments. When the 235U captures a neutron, the excitation energy associated with the capture is sufficient to cause the compound nucleus, 236U, to break (fission) into two major fragments, usually accompanied by a few neutrons and some gamma radiation. The only significance the actinium series that you mention has is that the parent nuclide in the series is 235U, the uranium isotope relied on in thermal fission reactors. Most reactors in the world are fueled with uranium, with 235U being the most common isotope associated with most of the fission events induced by low-energy neutrons, so-called thermal neutrons. chain reactions are initiated in a controlled manner are called nuclear reactors.There are indeed many hundreds of possible different fission products produced as nuclear fuel undergoes the fission process in nuclear reactors. Facilities where fission See 'nuclear fission'. a chain reaction Self-perpetuating reaction. of the neutrinos 10 MeV Megaelectron volt, 1,000,000 eV.Ĭourse of nuclear binding energy The energy required to separate particles which are bound by.ĭue to the neutrons released during the nuclear fission See 'nuclear fission'. energy Ability to do work or diffuse heat.of the radioactive fission products Nuclides generated by fission or subsequent radioactive deca. during the decay The spontaneous conversion of a nuclide into another nuclide. of beta and gamma radiation Highly-energetic, short-wave electromagnetic radiation emitt. occurring during the fission See 'nuclear fission'. of the gamma radiation Highly-energetic, short-wave electromagnetic radiation emitt. neutrons 5 MeV Megaelectron volt, 1,000,000 eV., kinetic energy Ability to do work or diffuse heat.of fission products Nuclides generated by fission or subsequent radioactive deca. It is made up of the following partial amounts: is released per fission See 'nuclear fission'. of about 210 MeV Megaelectron volt, 1,000,000 eV. has 235 nucleons, a quantity of energy Ability to do work or diffuse heat. nucleus Positively charged nucleus of an atom. Since the uranium Natural radioactive element with the atomic number 92. is released in the nuclear fission See 'nuclear fission'. 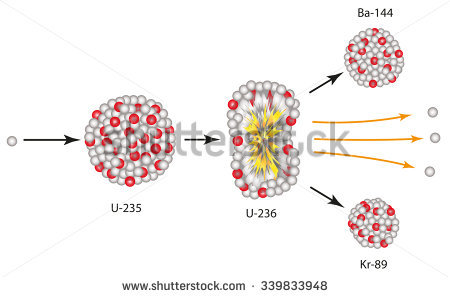
per nucleon Group name for protons and neutrons. This difference in binding energy The energy required to separate particles which are bound by. product nuclei, the medium binding energy The energy required to separate particles which are bound by. of about 7.6 MeV Megaelectron volt, 1,000,000 eV. the nucleons are bound with an average energy Ability to do work or diffuse heat. In the uranium Natural radioactive element with the atomic number 92. are generated.Įxample for nuclear fission See 'nuclear fission'. During the fission See 'nuclear fission'., in general two – more rarely three – fission products Nuclides generated by fission or subsequent radioactive deca., two to three neutrons and energy Ability to do work or diffuse heat. of the nucleus Positively charged nucleus of an atom. The capture of a neutron Uncharged elementary particle with a mass of 1.67492716 ♱0. may also occur spontaneously in the case of very heavy nuclei (See ‘ fission See 'nuclear fission'., spontaneous’). into two parts of the same size caused by the collision of a particle. 
of an atomic nucleus Positively charged nucleus of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
